Saudi Arabia In-Vitro Diagnostics Market Size and Forecast 2026–2034
Chronic Disease Burden, Vision 2030 Investments, and Diagnostic Innovation to Drive Steady Growth
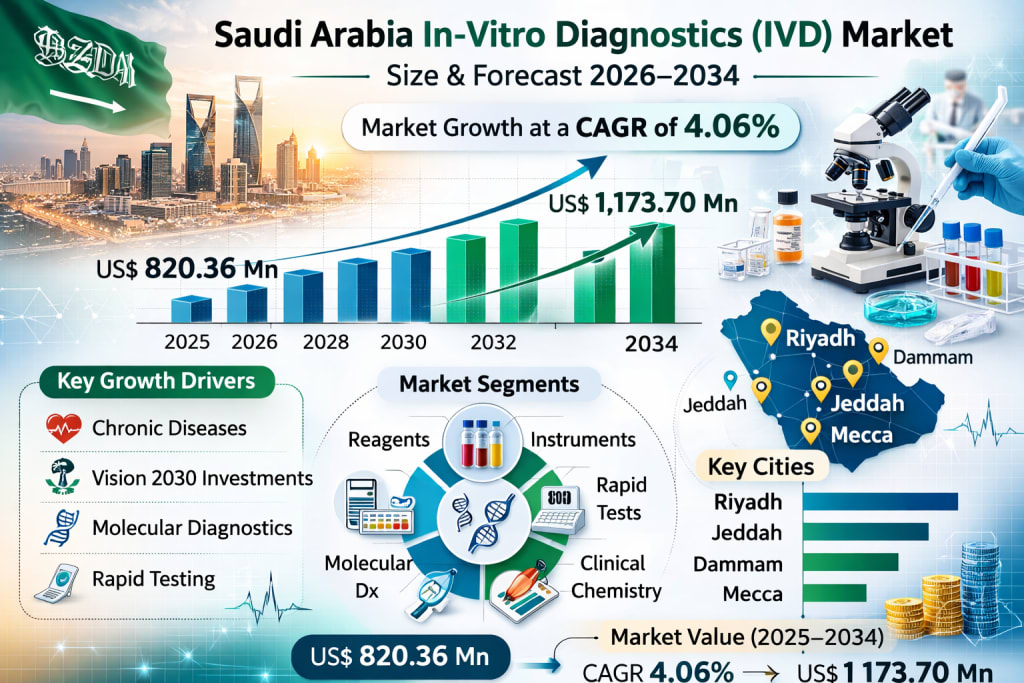
The Saudi Arabia in-vitro diagnostics (IVD) market is poised for steady and sustained expansion over the next decade. Driven by a rising burden of chronic and infectious diseases, growing healthcare expenditure, and a nationwide shift toward preventive care, the market is projected to grow from US$ 820.36 million in 2025 to US$ 1,173.70 million by 2034, registering a CAGR of 4.06% during 2026–2034.
This growth trajectory reflects not only increasing diagnostic volumes but also structural transformation under the Kingdom’s Vision 2030 agenda. From advanced molecular diagnostics to rapid point-of-care testing, Saudi Arabia’s diagnostic ecosystem is undergoing modernization, digitalization, and capacity expansion.
Saudi Arabia IVD Market Overview
In-vitro diagnostics refer to medical tests conducted outside the human body using samples such as blood, urine, tissue, or other bodily fluids. These tests play a critical role in disease detection, monitoring, prognosis, and treatment planning.
IVD products encompass:
Reagents and kits
Instruments and analyzers
Software and digital platforms
Automation systems
Molecular and immunoassay solutions
These technologies are widely used in clinical chemistry, hematology, immunology, microbiology, and molecular diagnostics.
In Saudi Arabia, IVD has gained widespread acceptance due to rising chronic disease prevalence, increased screening awareness, and government-backed healthcare transformation initiatives. Early detection and disease monitoring have become national priorities, significantly strengthening demand for advanced diagnostic platforms.
Growth Drivers of the Saudi Arabia IVD Market
1. Rising Prevalence of Chronic and Lifestyle-Related Diseases
Chronic diseases represent one of the largest health challenges in Saudi Arabia. Approximately 23% of the population is affected by diabetes, and nearly 25% suffer from hypertension. Cardiovascular disorders, obesity, and cancer incidence are also increasing, largely due to sedentary lifestyles and dietary habits.
These health trends directly translate into higher diagnostic demand. Chronic disease management requires:
Routine blood glucose testing
Lipid profiling
Kidney and liver function testing
Cardiac marker analysis
Hormonal monitoring
Regular diagnostic monitoring forms the backbone of long-term disease management. As awareness increases among both physicians and patients, diagnostic volumes continue to rise.
Furthermore, the emphasis on preventive screening has accelerated test utilization. Early detection programs for cancer and metabolic disorders are expanding across urban and semi-urban healthcare centers.
2. Government Healthcare Investments and Vision 2030
Saudi Arabia’s Vision 2030 framework has significantly reshaped the healthcare landscape. The government is investing heavily in:
Upgrading hospital infrastructure
Expanding diagnostic laboratories
Digitizing health records
Encouraging public-private partnerships
Attracting foreign healthcare investments
Major global health exhibitions and multi-billion-dollar healthcare agreements demonstrate the Kingdom’s ambition to become a regional healthcare hub.
Preventive care and early diagnosis are core pillars of the transformation strategy. This policy shift naturally increases demand for IVD technologies across public hospitals, private labs, and specialized diagnostic centers.
The expansion of private healthcare facilities has further boosted procurement of advanced analyzers, automated platforms, and molecular testing systems.
3. Technological Advancements and Molecular Diagnostics Expansion
Technological innovation remains central to IVD growth. Saudi Arabia is witnessing increasing adoption of:
PCR-based molecular testing
Chemiluminescent immunoassays (CLIA)
Automated ELISA systems
Point-of-care testing (POCT)
Digital lab integration systems
Molecular diagnostics are particularly transformative. They enable rapid detection of infectious agents, genetic mutations, and cancer biomarkers. With growing interest in precision medicine, demand for molecular testing platforms continues to expand.
Automation has also improved lab efficiency, reducing turnaround times while improving diagnostic accuracy. The integration of artificial intelligence and digital reporting tools is further strengthening diagnostic reliability.
Key Challenges in the Saudi Arabia IVD Market
1. High Cost of Advanced Technologies
Advanced molecular diagnostic systems and automated laboratory analyzers require significant capital investment. Smaller hospitals and regional diagnostic centers may struggle with:
Equipment procurement costs
Maintenance expenses
Training requirements
Software integration
Although large urban hospitals can afford high-end technologies, cost barriers may limit uniform adoption across rural areas.
Reimbursement frameworks for sophisticated diagnostics are still evolving, which can impact accessibility.
2. Dependence on Imports and Regulatory Complexity
Saudi Arabia’s IVD market remains heavily dependent on imported instruments, reagents, and kits. This creates exposure to:
Global supply chain disruptions
Currency fluctuations
Geopolitical risks
Shipment delays
Additionally, regulatory approval processes, while ensuring safety and quality, can delay the introduction of innovative diagnostic solutions. Manufacturers must navigate strict compliance requirements before market entry.
Segment Insights
ELISA & CLIA IVD Market
ELISA (Enzyme-Linked Immunosorbent Assay) and CLIA (Chemiluminescent Immunoassay) technologies are widely used for infectious diseases, hormonal disorders, autoimmune conditions, and oncology testing.
CLIA systems are increasingly preferred due to:
Higher sensitivity
Faster processing
Automation compatibility
Reduced manual intervention
Hospitals and centralized laboratories are rapidly upgrading to automated immunoassay platforms to meet growing test volumes.
Rapid Test Market
Rapid diagnostic tests have gained traction in emergency care, rural clinics, and decentralized healthcare settings. They are used for:
Infectious disease detection
Pregnancy testing
Blood glucose monitoring
Cardiac marker analysis
The COVID-19 pandemic accelerated awareness and infrastructure for rapid testing solutions. Government initiatives promoting accessibility and early detection further support growth in this segment.
IVD Reagents Market
Reagents represent a recurring revenue stream in the IVD ecosystem. Rising diagnostic volumes increase demand for high-quality reagents to ensure accuracy and consistency.
As automated analyzers become more common, reagent consumption naturally increases. Despite import dependency, reagents remain one of the strongest growth contributors within the Saudi IVD market.
Clinical Chemistry Market
Clinical chemistry testing dominates routine diagnostics in Saudi Arabia. Tests include:
Blood glucose
Lipid profiles
Electrolytes
Liver function tests
Kidney function panels
Given the high prevalence of diabetes and cardiovascular disorders, clinical chemistry testing volumes are expected to remain robust throughout the forecast period.
Automation adoption in hospitals further strengthens this segment’s outlook.
Microbiology IVD Market
Microbiology testing plays a vital role in detecting bacterial, viral, and fungal infections. Growing awareness of antimicrobial resistance and hospital-acquired infections has increased demand for:
Automated culture systems
Rapid pathogen identification tools
Molecular microbiology platforms
Infection control initiatives and public health surveillance programs are key growth drivers.
City-Level Market Dynamics
Riyadh
As the capital city, Riyadh represents the most advanced IVD market in the Kingdom. Government healthcare investments, high hospital density, and advanced diagnostic adoption make it the largest regional contributor.
Chronic disease prevalence and oncology screening programs further stimulate demand.
Jeddah
Jeddah benefits from a strong private healthcare sector and medical tourism. Its strategic port access facilitates medical imports. The city shows rising adoption of rapid and molecular diagnostics.
Mecca
Seasonal pilgrimages significantly increase temporary population density. Demand for infectious disease testing, rapid diagnostics, and emergency healthcare services spikes during Hajj and Umrah seasons.
Dhahran
Dhahran’s market is supported by corporate healthcare and expatriate populations. While smaller in volume, it maintains consistent growth due to high healthcare standards and insurance coverage.
Competitive Landscape
The Saudi Arabia IVD market is characterized by the presence of leading global diagnostic companies, including:
Roche Diagnostics
Danaher Corporation
Abbott Laboratories
Thermo Fisher Scientific
Bio-Rad Laboratories, Inc.
Sysmex Corporation
Becton Dickinson
bioMérieux
These companies compete across immunochemistry, molecular diagnostics, hematology, microbiology, and clinical chemistry platforms. Strategic partnerships, localization efforts, and product innovation remain central to competitive positioning.
Market Segmentation
Test Types:
ELISA & CLIA, PCR, Rapid Test, FIA, In Situ Hybridization, TMA, Sequencing, RIA, Isothermal Amplification, Others
Products:
Reagents, Instruments, Services
Applications:
Immunochemistry, Clinical Chemistry, Hematology, Coagulation, Molecular Diagnostics, Microbiology
Cities:
Riyadh, Jeddah, Dammam, Dhahran, Jubail, Khobar, Tabuk, Mecca, Medina, Rest of Cities
Final Thoughts
The Saudi Arabia in-vitro diagnostics market is positioned for stable and strategic expansion through 2034. With a projected rise from US$ 820.36 million in 2025 to US$ 1,173.70 million by 2034 at a CAGR of 4.06%, the sector reflects both epidemiological need and structural healthcare transformation.
Chronic disease prevalence, preventive screening programs, and Vision 2030 healthcare investments will continue driving diagnostic volumes. At the same time, technological innovation—particularly in molecular diagnostics and automation—will redefine testing standards across the Kingdom.
While challenges such as import dependency and cost barriers persist, Saudi Arabia’s commitment to healthcare modernization ensures that in-vitro diagnostics will remain a cornerstone of the nation’s medical ecosystem in the years ahead.






Comments
There are no comments for this story
Be the first to respond and start the conversation.